
Understanding Formulation Stability: Microtrac Solutions For Chemical And Physical Analysis
Formulation stability is one of the cornerstones of successful product development. Whether in pharmaceuticals, cosmetics, food, or agrochemicals, every industry faces the same challenge: ensuring that formulations remain stable over time.
In broad terms, stability determines how long a product lasts on the shelf and whether it performs safely and effectively. Both physical and chemical factors influence this, and both require careful testing from early R&D through large-scale manufacturing.
The meaning of formulations goes far beyond mixing ingredients. In formulation chemistry, every excipient, stabilizer, or preservative plays a role in protecting performance and extending shelf life.
Examples of formulations include oral drug suspensions, cosmetic creams, nutrient-fortified beverages, and pesticide sprays, all of which need strict stability evaluation before reaching consumers.
What Is Chemical Stability?
When scientists talk about stability, one of the first questions is: is the formulation chemically stable?
To define chemical stability, we look at whether the active compounds in a product remain intact over time. A chemically stable compound resists degradation, meaning its molecular structure and potency do not change during manufacturing, storage, or normal use.
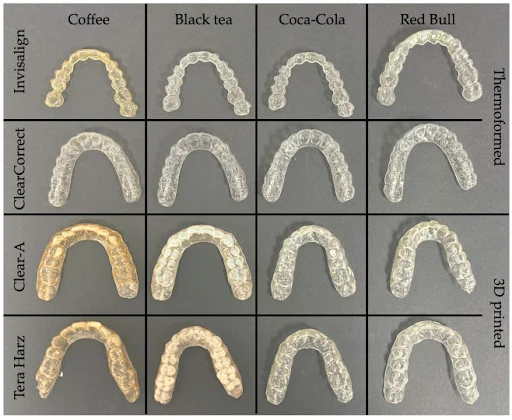
Aligners after 24 h of immersion
Loss of chemical stability can happen through several pathways: oxidation, hydrolysis, pH shifts, or microbial activity. Each reaction may produce impurities, reduce effectiveness, or even create safety concerns. For this reason, chemical stability testing is a cornerstone of formulations chemistry, determining whether a compound can be used in pharmaceutical formulations, cosmetic creams, agrochemical sprays, or food products.
Ultimately, chemical stability sets the limit for a product’s shelf life. If a formulation cannot maintain its intended chemical profile, it cannot guarantee performance, safety, or compliance once it reaches the end user.
Distinguishing Physical vs. Chemical Stability
Formulation stability has two dimensions: physical and chemical. While both are essential, they stem from different mechanisms and require distinct testing methods.
Aspect | Physical Stability | Chemical Stability |
Definition | Ability of a formulation to maintain appearance, texture, viscosity, and particle size over time | Ability of compounds to remain chemically intact, preserving molecular structure and potency |
Visible Signs of Instability | Sedimentation, creaming, caking, phase separation, texture change | Loss of potency, formation of degradation products, changes in color or odor |
Main Causes | Environmental factors: temperature fluctuations, humidity, light exposure, mechanical stress | Molecular reactions: oxidation, hydrolysis, microbial contamination, pH shifts |
Impact on Product | Reduced performance, compromised user experience, shorter shelf life | Reduced efficacy, safety risks, regulatory failure, shortened shelf life |
Testing Focus | Particle size analysis, rheology, visual inspection, accelerated aging | Chromatography (HPLC, GC), mass spectrometry, FTIR, long-term chemical stability studies |
Understanding these distinctions allows researchers to design more targeted testing strategies, ensuring both the appearance and function of formulations remain stable throughout their intended shelf life.
Factors Affecting Formulation Stability
Both physical stability and chemical stability can be compromised by external conditions. Understanding these influences helps researchers design more robust formulations and predict product performance throughout its shelf life.
Factors impacting physical stability include
- Temperature changes – may cause phase separation, caking, or changes in viscosity.
- Humidity – accelerates particle agglomeration, swelling, or precipitation in moisture-sensitive systems.
- Light exposure – can lead to discoloration or photo-induced breakdown of appearance and texture.
- Mechanical stress – shaking, transport, or handling can destabilize suspensions or emulsions.
Factors impacting chemical stability include
- pH shifts – affect solubility and can trigger chemical reactions.
- Oxygen exposure – promotes oxidation, leading to degradation and impurity formation.
- Hydrolysis – breakdown of chemical bonds in the presence of water, common in pharmaceutical formulations.
- Microbial contamination – introduces biochemical reactions that compromise safety and shorten stability.
By controlling these variables during development, storage, and distribution, companies can significantly improve the reliability of stable chemicals used in modern formulations.
Techniques For Measuring Stability
Accurate testing is the backbone of formulation chemistry, helping scientists confirm whether products remain reliable throughout their expected shelf life. Since physical and chemical stability are driven by different mechanisms, they require complementary methods of analysis.
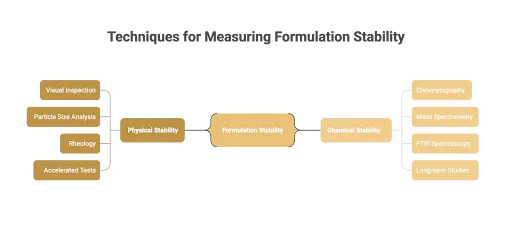
Measuring Physical Stability
Physical testing focuses on the visible and structural changes within a formulation. Early signs like sedimentation, creaming, or phase separation are often detected through visual inspection.
To go deeper, techniques such as particle size analysis monitor how particles evolve in suspension, while rheological measurements track viscosity and flow behavior under stress. In many industries, accelerated stability testing is also used, where heat or agitation simulates long-term conditions to predict performance faster.
Measuring Chemical Stability
Chemical testing evaluates whether the molecular structure of active compounds remains intact. Tools such as chromatography (HPLC, GC) separate and identify components, revealing degradation pathways.
Mass spectrometry (MS) provides precise molecular weight information to detect impurities, while FTIR spectroscopy highlights changes in molecular bonds. Long-term controlled studies remain the gold standard for confirming whether stable chemicals will maintain their properties across real-world storage conditions.
Together, these approaches provide a complete picture of how formulations behave, ensuring that every compound used in pharmaceutical, cosmetic, food, or agrochemical products meets both performance and safety requirements.
Microtrac Solutions For Formulation Stability
Accurately measuring formulation stability requires tools that deliver reliable, reproducible results. Microtrac’s portfolio of particle size analyzers and stability instruments is designed to meet the diverse needs of industries working with pharmaceuticals, cosmetics, foods, and agrochemicals.
- Particle size analyzers provide detailed insight into dispersion quality, helping predict physical stability before visible separation occurs.
- Rheological and dynamic analysis tools assess flow behavior, texture, and aging, offering a deeper understanding of performance under stress.
- Chemical analysis support integrates with chromatographic and spectroscopic methods, ensuring that both molecular integrity and physical properties are monitored side by side.
By combining these capabilities, Microtrac enables scientists and manufacturers to design more stable formulations, reduce development time, and safeguard product shelf life.
The Role of TURBISCAN Technology
Within Microtrac’s portfolio, the TURBISCAN series is recognized as the global standard for analyzing formulation stability. Using Static Multiple Light Scattering (SMLS), it identifies early signs of destabilization, such as aggregation, separation, or aging, long before they become visible.

Buy the STABILITY ANALYZER TURBISCAN LAB with a free professional consultation and equipment financing today
What sets TURBISCAN apart is its ability to:
- Measure shelf-life behavior in hours instead of weeks
- Assess dispersibility and redispersion after stress or storage
- Operate across a 20–60 °C temperature range to simulate real conditions
The series includes three models tailored to different applications: the TURBISCAN TOWER for high-throughput workflows, the TURBISCAN TRILAB for flexible R&D and QC tasks, and the TURBISCAN LAB, widely regarded as the global benchmark in formulation chemistry.
By delivering fast, quantitative results without requiring expert operators, TURBISCAN helps teams accelerate development, improve product reliability, and ensure longer, more predictable shelf life.
Benefits of Using Microtrac Instruments
For many labs, stability testing can feel like a waiting game, weeks of observation, trial-and-error methods, and inconclusive results. Microtrac instruments change that dynamic by giving researchers tools to see what’s happening inside a formulation in real time.
Instead of relying on visual checks or broad assumptions, teams can track the smallest shifts in particle size, bonding, or dispersibility. This precision allows them to catch problems early, fine-tune formulations faster, and avoid wasting material on unstable designs.
The benefits go beyond speed:
- Early insight for R&D – Detect instability within hours, guiding smarter formulation choices from the start.
- Stronger QC decisions – Quantify stability with reproducible data, not subjective observations.
- Confidence in market release – Demonstrate that products will keep their integrity and shelf life under real-world conditions.
For companies where performance and safety depend on stable chemicals, whether it’s a new oral drug, a cosmetic emulsion, or a fortified beverage, Microtrac instruments provide the clarity needed to move forward with certainty.
How Additive Plus Guides Your Instrument Selection
Selecting instruments for stability testing can be complex. As a provider of Microtrac solutions in the U.S., Additive Plus helps companies match their needs with the right tools.
From particle size analyzers to the TURBISCAN series, we guide clients in choosing instruments that support reliable data, stronger formulations, and proven shelf life. Our team understands the challenges of working across pharmaceuticals, cosmetics, food, and agrochemicals, and helps align testing methods with real-world application goals.
With Additive Plus, you gain direct access to Microtrac’s portfolio along with expert guidance, so every investment in stability testing translates into better outcomes for your products and your customers.
Ready to take the next step in improving your formulation stability testing?
FAQ
What is the chemical stability definition in simple terms?
Chemical stability refers to a compound’s ability to resist chemical change over time. A chemically stable product maintains its molecular structure and potency under storage and use, ensuring predictable performance, safety, and compliance throughout its shelf life.
What are formulations, and why are they important?
Formulations are combinations of active ingredients with excipients designed to deliver consistent performance. In formulation chemistry, the choice of stabilizers, solvents, and preservatives determines whether a product will remain effective. Understanding the meaning of formulations helps ensure reliability across pharmaceuticals, cosmetics, foods, and agrochemicals.
Can this compound be used in pharmaceutical formulations?
Not every compound is suitable for pharmaceutical formulations. Stability testing confirms whether the active ingredient remains intact under conditions like temperature, pH, and oxygen exposure. If a compound lacks chemical stability, it may degrade quickly and cannot be used safely in drug products.
What are examples of formulations outside pharmaceuticals?
Examples of formulations include cosmetic emulsions, fortified drinks, agrochemical sprays, and industrial coatings. Each requires both physical stability and chemical stability to meet quality standards. By testing stability, companies ensure these formulations remain effective from manufacturing to the end of their shelf life.
How is shelf life determined in stability studies?
Shelf life is established by exposing products to controlled environmental conditions, temperature, humidity, and light, and monitoring changes in physical and chemical properties. If a product remains within safe, functional limits, it is considered chemically stable and assigned a verified shelf life for distribution and use.
Related Posts
Ashkhen Ovsepyan
Additive manufacturing expert and tech business leader, focused on global strategy, innovation, and advancing women in 3D printing.




